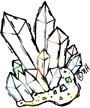
 Crystals are solids with particles arranged in a regular, repeating pattern with flat surfaces. Most solids are crystalline solids. The crystal diagram shown is of quartz, which is made of silicon dioxide, the same chemical found in sand. Without any impurities, quartz is colorless and transparent. Impurities, such as iron, give quartz crystals a pink color. The color is distinctive and the crystal is called pink quartz.
Crystals are solids with particles arranged in a regular, repeating pattern with flat surfaces. Most solids are crystalline solids. The crystal diagram shown is of quartz, which is made of silicon dioxide, the same chemical found in sand. Without any impurities, quartz is colorless and transparent. Impurities, such as iron, give quartz crystals a pink color. The color is distinctive and the crystal is called pink quartz.
Ice is a crystal made of molecules of water. A large piece of ice is made up of many small ice crystals that fit together like a puzzle. Ice has a milky color when air is trapped in the crystal. Glaciers have a distinctive blue color not because of impurities but like the sky, some of the colors of light making up sunlight is absorbed by the ice and the blue colors are reflected or transmitted (pass through). The photo is of me on the top of a glacier in Alaska. The shades were necessary to pro tect my eyes from the sunlight reflecting off of the ice. While they protected my eyes, without sunscreen on my face, I would have looked like a negative image of a raccoon– brown skin with white rings around my eyes.
tect my eyes from the sunlight reflecting off of the ice. While they protected my eyes, without sunscreen on my face, I would have looked like a negative image of a raccoon– brown skin with white rings around my eyes.
The shape of crystals depends on the arrangements of the particles within it. A unit cell is the smallest group of particles within a crystal that retains the geometric shape of the crystal. For example, the unit cell of halite (table salt crystals) are cubic (box-shaped). For halite crystals to form, the cubic cells must be able to move about so that they can be positioned with their faces (flat surfaces) against one another.
I have researched different parts of the world–from the geographic south pole to Barrow Alaska, on the coast of the Arctic Ocean. Our planet has so many wonderful sites. For information about some of the Earth’s biomes, see Janice VanCleave’s Science Around the World.
Science Around the World
This book presents interesting facts and fun experiments that relate to the different geographical regions of the world-also known as biomes. Each different biome-forest, grassland, desert, and tundra-has its own unique plant life, animal life, and climate. The experiments, activities, and facts in this book help explain how the different biomes work and show the importance biomes play in keeping life on Earth so fascinatingly diverse.
(Paid Link)
