 Sample Worksheet
Sample Worksheet
Teaching Tips
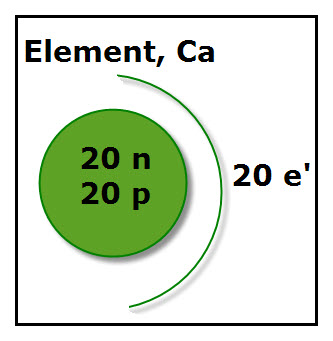
1. The diagram gives just enough information to answer the following questions if the students are familiar with these terms:
atomic number, electrons, mass number neutrons, nucleon, nucleus, protons,
2. Each sample question is followed by its answer.
Instructions: Using the diagram fill in the blanks for each statement.
1. The name of the element’s nucleons are __________ and _________.
(Nucleons are the particles inside the nucleus of an atom. The green circle represents the nucleus. The two nucleons are neutrons and protons.)
2. What is the name of the element? _________ (Ca is the symbol for calcium.)
3. What is the atomic number of the element? ___________(The atomic number of any element is equal to the number of protons inside the nucleus of each of the element’s atoms. For Ca, there are 20 protons, thus the atomic number for Ca is 20.)
4. What is the mass number of the atom shown? _________ (The mass number is the sum of the number of nucleons of the atom. For the Calcium atom, there are 20 neutrons + 20 protons, thus its mass number is 40.)
Big Book of Science Experiments
A book of fun informative experiments about astronomy, biology, chemistry, earth science, and physics.
(Paid Link)
