Question: What are atoms made of?
Answer: Atoms are the smallest part of an element (single substances, such as copper, gold, and carbon).
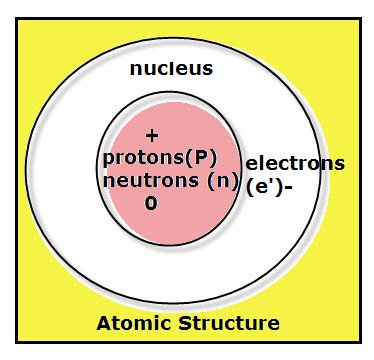
Atoms can be divided into two basic regions:
1. The region in the center of an atom is called the nucleus. Particles inside the nucleus are called nucleons, such as protons and neutrons.
- Protons are positively charged particles.
- Neutrons are particles without a charge.
2. The region outside the nucleus is where the atom’s electrons move.
While there are no real boundaries that hold nucleons inside the nucleus or to contain electrons within a certain distance from the nucleus, the atomic particles remain in certain areas.
Electrons farthermost from the nucleus are more easily rubbed off—pulled away by another atom–or shared with another atom.
Each of the 101 chemistry experiments has a purpose, list of materials, step-by-step instructions and illustrations, expected results, and a science explanation in understandable terms.
(paid link)
