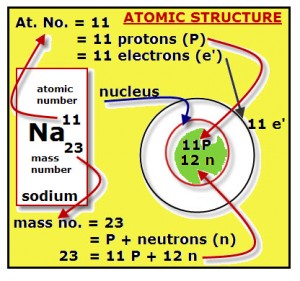
ATOMIC STRUCTURE OF SODIUM
The Periodic Table provides the information about every known element. In the diagram, one cell from the periodic table is shown. This is the cell for the sodium element. The contents of the cell shown are:
Name: sodium
Symbol: Na
Atomic Number: 11
Mass Number: 23
Study the example diagram, and then fill in the blanks to describe the procedure for drawing an atomic structure for sodium.
Facts:
1. The atomic number = the number of protons
2.The atomic number = the number of electrons
3. The mass number = the number of protons + the number of neutrons
4. Nucleons = the particles inside the nucleus.
Instructions for Drawing the Atomic Structure of Carbon
1. Draw two concentric circles (one inside the other). The inside smaller circle is called the ________________. Label this circle.
2. The nucleus contains nucleons. The names of these nucleons are ____________ and _________________.
3. The outer circle represent the region outside the nucleus. This region contains particles called __________, which have a ___________charge. Write the symbol and number of these particles on the outer circle.
4. The number of protons that an atom has is called the atom’s _____ ______. Write the symbol and number of these particles inside the nucleus.
5. The number of neutrons inside the nucleus is equal to the atom’s _____ ______ minus the atom’s number of __________. Write the symbol and number of neutrons for sodium inside the small circle, the nucleus.
6. The number of electrons outside the nucleus of an atom is equal to the number of _________inside the nucleus of the atom.
Big Book of Science Experiments
A book of fun informative experiments about astronomy, biology, chemistry, earth science, and physics.
(Paid Link)