Conduction (or heat conduction) is the transfer of thermal energy between neighboring molecules in a substance due to differences in temperature.
Thermal energy is always transferred from a region of higher temperature to a region of lower temperature.
The transfer of thermal energy continues until each region has the same temperature.
Heat Conduction takes place in solids, liquids, and gases, but works best in materials that have simple molecules that are located close to each other. For example, metals are better conductor than wood or plastic.
Heat Conduction is the transfer of energy from one molecule to another. This energy transfer occurs when molecules hit against each other.
The pool balls shown model the transfer of energy from one molecule to another. The pool balls, being molecules. The orange ball in motion has more energy than the black stationary ball. When the moving orange ball collides with the stationary black ball, there is a transfer of energy from the orange ball to the black ball. The state of motion of the two balls after the collision depends on the amount of energy transferred as well as other variables, including the mass of the balls.
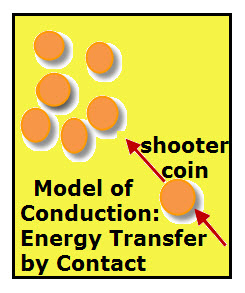
Coins, like the pool balls, can be used to model Heat Conduction.
1. Randomly place 6 or more pennies (any coin of the same denomination or metal washer will work) on a flat, smooth surface. The coins represent atoms or molecules an substance is made of.
2. Place a single penny about 6 inches from the group of coins. This single coin will be the “shooter.”
3. With your finger, thump the “shooter” coin toward the group of coins. The shooter coin now represent an atom or molecule with more kinetic energy than do those in the group.
4. Observe any motion of the coins in the group as well as any change in motion of the shooter coin. Changes in motion of any of the coins represents a transfer of energy from the shooter coin.
Changes in motion of any of the coins represents a transfer of energy from the shooter coin.
A. How many group coins moved?
B. Thumping the shooter coin causes it to accelerate in a direction toward the group of coins.What affect does the collision have on the shooter coins acceleration and direction of motion?