This Unit on the Periodic Table is being developed. In other words, I am writing the activities in response to your feed back. Please read the activities and let me know how they can be changed to make them more useful. When applicable, I will use your questions. For example:
Question: Why is the periodic table so important?
Answer:
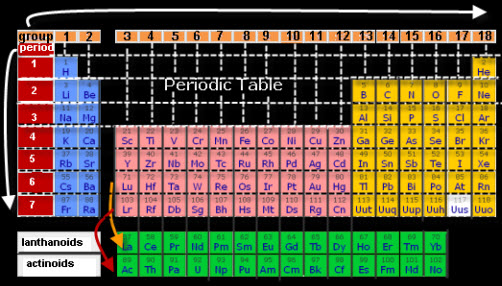
The periodic table provides an abundant amount of information about each element, such as whether the element is a metal or non-metal as well as how active it is. The table also tell you which elements are most likely to chemically combine to form compounds, such as the combination of these elements:
hydrogen + oxygen ———–> water
sodium + chlorine ————->table salt
iron + oxygen ——————> rust
For example:
H (hydrogen) in group 1, row 1 burns very quickly. Blimps at one time were filled with hydrogen because it is the lightest gas. The Hindenburg blimp was was filled with hydrogen and exploded soon after it landed.
He (helium) in group 18, row 1 is very inactive. It is the gas used in balloon so that they float in air.
The exciting news about the periodic table is that it is a tool to use, not to memorize unless you just want to.
Let’s Get Started
Activity 1: Elements
Question: What is an element?
Answer:
An element is a substance made of the same kind of atoms. For example, the element gold, is made of atoms of gold, and the element carbon is made of atoms of aluminum.
Did You Know?
A diamond and coal are both examples of the carbon element. They look different because of how the carbon atoms are linked (bonded) together.
The list on the right contain the first 36 elements on the periodic table. Do you see any element names that you are familiar with? How about oxygen? Take a deep breath. About 20 percent of the air that you inhaled is oxygen gas. Have you ever used aluminum foil? It is made of aluminum atoms, so it is an example of an element.
Activity 1: Learning the Names of Elements.
Materials
36 index cards
Procedure:
1. Print the names of the elements in the list on the cards, one name per card.
2. Divide the cards into two stacks, and make note of how many cards are in each stack. You will repeat this again after you complete several periodic table activities.
Stack 1: Elements that you know something about.
Stack 2: Elements that you do not know anything about.
Activity 2: Learning About the Symbols for Elements
Materials
36 index cards (new cards)
36 index cards from Activity 1 (names of elements)
Facts:
A symbol is used to represent something. The symbol for most elements is one or two letters. For those with two letters, the first letter is capitalized and the second is lower case.
Procedure:
1. Using the periodic chart found HERE ,
for information about elements 1-36.
2. Print one symbol on each index card.
2. Pair up the two sets of cards. Each pair contains the elements name and symbol.
Any ideas for using these cards to play a game?
Activity 3: Learning the Location of Elements on the Periodic Table
Materials
36 index cards (new cards)
36 index cards from Activity 1 (names of elements)
36 index cards from Activity 2 (symbols of elements)
Procedure:
1. Using the periodic chart found HERE , find the location code for each of the first 36 elements on the periodic chart.
Note: The Location Code has two number, the group number first, and then the row number.
For example:
Na, sodium is in a box where group 1 and row 3 meet.
2. Print one symbol on each card.
3. Group the three sets of cards. Each group should contain 3 cards.
Card 1: Element Name
Card 2: Element Symbol
Card 3: Element Location Code
Big Book of Science Experiments
A book of fun informative experiments about astronomy, biology, chemistry, earth science, and physics.
(Paid Link)