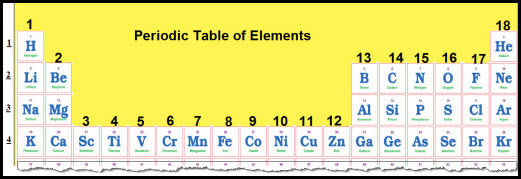
 The Periodic Table of Elements is arranged so that elements in groups have similar properties. The valence electrons for Groups 1, 2, 13, 14, 15, 16, 17, and 18 are easily to determine.
The Periodic Table of Elements is arranged so that elements in groups have similar properties. The valence electrons for Groups 1, 2, 13, 14, 15, 16, 17, and 18 are easily to determine.
Groups 1 and 2
The valence electrons for Groups 1 and Group 2 are equal to the group number.In other words, Valence Electrons for Group 1 =1, and Valence Electrons for Group 2 =2.
Valence Electrons: The electrons in the outermost energy level of an atom.
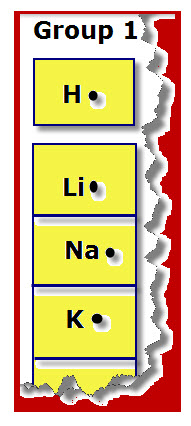
Group 1: The elements for Group 1 shown in the diagram are H, Li, Na, and K. These four elements as well as the other elements in this group have ONE valence electron.
Dot Diagrams show the symbol of an element with dots, representing valence electrons, positioned around the symbol.
Group 2: The elements for Group 2 shown in the diagram ar e Be, Mg, Ca, and Sr. These four elements as well as the other elements in Group 2 have TWO valence electron.
e Be, Mg, Ca, and Sr. These four elements as well as the other elements in Group 2 have TWO valence electron.
The diagram shows the dot diagrams for Be, Mg, Ca, and Sr.
Groups 13-18
To predict the valence electrons for groups 13 through 18:
Subtract 10 from the group number.
The answer is equal to the number of valence electrons for each element in the group.
 |
Chemistry For Every Kid |