Chemistry Facts:
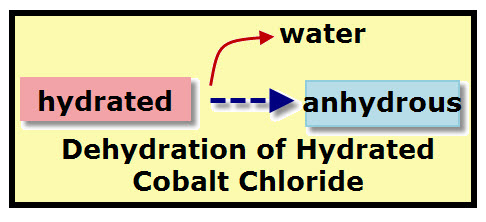
Hydrates are chemicals that have water as part of their structure. If these chemical lose some or all of this water they change in appearance.
- Cobalt Chloride is an example of a chemical hydrate.
When cobalt chloride is hydrated (having water) its color is pink.
When cobalt chloride is dehydrated (loses water) its color is blue. -
Hydrated cobalt chloride has this chemical formula: CoCl2•6H2O
-
Dehydrated cobalt Chloride has this chemical formula: CoCl2.
Dehydrated cobalt chloride is called anhydrous cobalt chloride.
For home study and in a classroom, I recommend using cobalt chloride hydrate paper. The paper is much easier to work with, less expensive, and most of all safer. Hydrated cobalt chloride paper is pink. When the paper dries, its color changes to blue. The hydrated paper can be placed in a warm area so that the water evaporates. If the humidity of the air is very dry, the paper will dry. If the humidity is high, the paper absorbs water from the air and turns pink.
Cobalt chloride paper can be used to determine changes in humidity. Thus, cobalt chloride paper can be used as a humidity gage.
For more information about ordering copper chloride hydrate paper strips, click on the puffy cloud or search for where to purchase cobalt chloride paper.
Each of the 101 chemistry experiments has a purpose, list of materials, step-by-step instructions and illustrations, expected results, and a science explanation in understandable terms.
(paid link)