How Can Water Freeze and Melt at the Same Temperature?
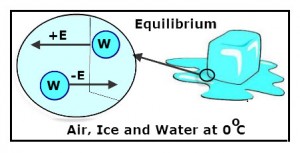
The freezing/melting point of water is OºC. But it is not an instantaneous physical change. Instead, both liquid water and ice can be present at OºC. At this temperature, there is a constant exchange of water molecules between the ice and liquid water as shown in the diagram.
Note: The change of the state of matter from a solid to a liquid or vice verse, does not change the chemical composition of the matter. Instead, there is a change in energy which affects how the particles in the matter move and their location to one another.
The letter “E” represents energy. Notice that frozen water molecules(ice) must gain energy (+E) to melt and liquid water molecules must lose the same amount of energy (-E) to freeze.
As long as the air, liquid water and ice are all at the freezing/melting temperature of water, the number of liquid water molecules that freeze are equal to the number of ice molecules that melt. Thus, there is no noticeable change in the amount of ice or water. Also, there is no temperature change because the amount of energy gained equals the amount of energy lost. The system is said to be at equilibrium.
Cold is the loss of heat energy. So a decrease in temperature is due to movement of thermal energy from a warmer substance to a cooler substance. Heat is the transfer of thermal energy.
Each of the 101 chemistry experiments has a purpose, list of materials, step-by-step instructions and illustrations, expected results, and a science explanation in understandable terms.
(paid link)
