The spy pen contains ink that is invisible, which means that under ordinary lighting the ink does not reflect visible light to your eyes.
Visible light is one of seven different types of light energy (also called electromagnetic energy) and is the only type of light that you can see.
Visible light is a band of light energy composed of different amounts of energy that are perceived as being different colors. These colors are visible in rainbows and listed from the least energy to the most energy are, red, orange, yellow, green, blue, indigo, and violet. The mnemonic ROY G. Biv is used to help remember the colors in order.
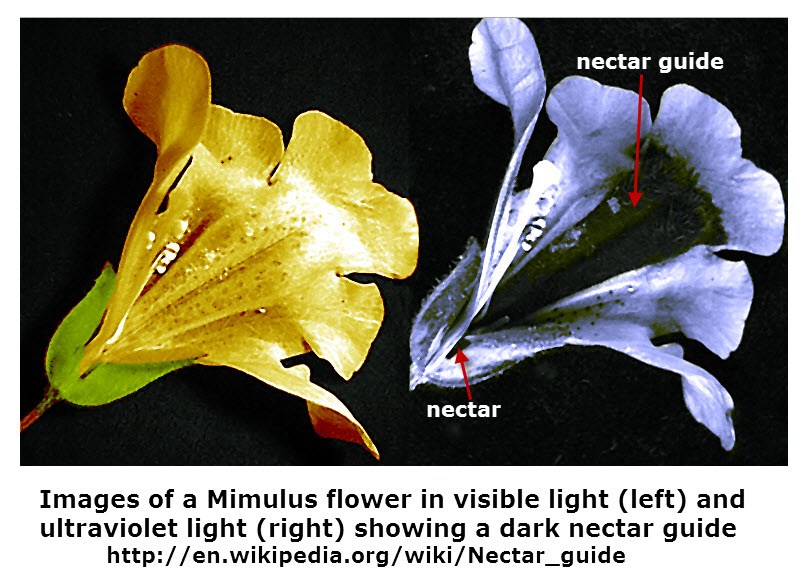
Ultraviolet (UV) light is not part of the visible light band, thus you cannot see most materials that are illuminated by UV light. For example, there are markings on flowers that reflect UV light that you cannot see, but is visible to bees and other insects that are able to see UV light. The picture shows how you view the flower and how is it viewed by bees looking for nectar. (How do scientists know what bees are able to see? I am asking the same question. Will be researching this. Let me know if you know the answer.)
What Makes the Spy Pen Ink Only Visible in UV Light?
The answer is the presence of a phosphor in the ink. Phosphors are chemical substance that absorbs ultraviolet light and emits visible light.
FYI: Phosphors may but do not generally contain the phosphorus element.
When UV light shines on a phosphor it causes electrons in the phosphor atoms to be energized. The energized electrons release photons, which are bundles of light energy and return to their natural unenerized state.
Using a simple drawing of an atom, you can visualize how high energy UV light can cause an atom to emit lower energy visible light.
First, know that light energy can be described as small packets called photons. A photon of UV light has more energy than does a photon of visible light. Colors that you see, such as a green leaf, is due to photons of a specific amount of energy being reflected from the leaf and entering your eyes. The color of the invisible ink in the Spy Pen depends on the energy of the visible light photons the ink emits when UV light shines on it. The diagram shown using a green circle to represent the nucleus of a phosphor atom. The circles around the nucleus are energy levels or distances from the nucleus where the electron shown is most likely to be found in. Remember the diagram is 2-D and atoms are 3-D. So the electron would be zipping around within a sphere or cloud around the the nucleus. Ground state is the area the electron is most likely to be in under normal conditions. Photons are packet of light energy. Photons of UV light have more energy than do photons of visible light. When UV light shines on the invisible ink from the Spy Pen, the electrons in the phosphors within the ink absorb photons of UV light. Electrons are within certain energy levels, clouds surrounding the nucleus of an atom, because of the amount of energy they have. An electrons that absorbs high energy photons is said to be energized and moves farther away from the nucleus. Energized Electrons are also called excited electrons. Energized electrons emit photons of visible light and move back to their ground state. Fluorescence is when electrons are energized by high energy photons and the energized electrons emit lower energy visible photons. Other Examples of Fluorescence 1. The exoskeleton of scorpions contain phosphors. At night, scorpions can be located using a black light. The UV radiation emitted by this light causes the scorpion to fluoresce (absorb UV photons and emit visible light photons). 2. Spy Pens with invisible Ink can be purchased at www.Teachersource.com For information about these pens see on this website Spy Pens
Add Rigor: Steganography (![]() ) is the art and science of encoding hidden messages in such a way that no one, apart from the sender and intended recipient, suspects the existence of the message.
) is the art and science of encoding hidden messages in such a way that no one, apart from the sender and intended recipient, suspects the existence of the message.
Activity: Make your own invisible ink that emits visible light when viewed under UV light from your Spy Pen or from UV flashlight found at www.Teachersource.com
Big Book of Science Experiments
A book of fun informative experiments about astronomy, biology, chemistry, earth science, and physics.
(Paid Link)