Earth’s only inexhaustible energy source comes from the Sun. The supply of chemicals on Earth are fixed, unless you count meteorites from space. The food you eat is made of chemicals–where did they come from? If you trace the source of each chemical in your body, you would end up going in circles. This is […]
Science Fair Projects: Question
I love my apple slicer. Like the apple slices in the photo, I press the slicer into an apple and voila’ I have nice slices that spread out like flower petals. Even the apple core makes a decorative center for apple “petals.” But, the apple slices must be eaten in a short period of […]
Periodic Table
The periodic table is something that even young children can study and have fun putting all the little boxes together. The table on this page is reduced and part of its bottom sections are not shown. Young learners can make Element Flash cards showing the atomic number, symbol, and element name on one side of […]
Elements
Notes from Janice’s Desk A friend tweeted the link to the element video below. I love it. It is a fun, colorful, musical production about elements, which is exactly the way that chemistry or any science topic should be introduced. Below the video is an element book by Tom Jackson. It is a must see. […]
Radiation
Today, while looking for a book, I found a sack of Ultraviolet Detecting Beads.These beads absorb UV radiation and change from white to different colors depending on the dye molecule in the bead. How cool is that? Of course I had to play with the bead to find out what colors I had left in […]
Chemistry:Corrosion
Notes from Janice’s Desk Lately there have been several water leaks in my house. One was due to an old water heater that has not been used for years. I must admit that the beautifully colored crusted areas on the pipes caught my eye. If you look close you can see dark blue crystals. While […]
Chemistry: Solutions
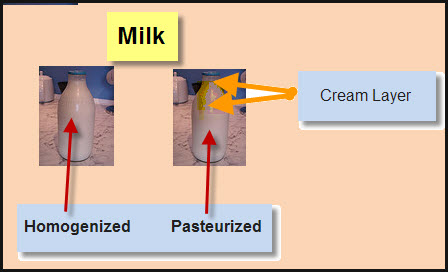
Notes from Janice’s Desk When I was a kid milk was pasteurized. At the time I didn’t understand that pasteurized meant the milk was heated to kill any bacteria. In fact I never thought about the word until a new kind of milk was introduce. This new stuff was call homogenized and my parents were not […]
Chemistry
Notes from Janice’s Desk Is chemistry gumdrops and toothpick models? Yes! Is chemistry mysterious bubbling flasks? Yes! Is chemistry explosions? Yes! Chemistry is all of the above and lots more. In fact, chemistry is part of everything that is fun–from making silly putty to a fancy birthday cake. So it is a great topic for […]
Physical Properties
Notes from Janice’s Desk Physical properties describe the characteristics of a substance or any bit of matter. There are two types of physical properties: One type of physical property DOES NOT depend on the amount of matter present. These include color, texture,shape, smell, state of matter (solid, liquid, and gas), sound, and taste. A second […]
Tips for Teaching Chemistry
Have you ever used art to teach chemistry? I have! In fact once I used special paper that changed color with changes in the humidity (amount of moisture in the air.) The paper was blue when the air was dry and pink when the humidity increased due to an increase in the moisture in the […]