This photo by Sara Robinson, captures my chocolate fantasy. Being diabetic, chocolate candy, chocolate icing, chocolate etc… for me can only be a part of my dreams. Studies have shown a relationship to chocolate and mood. STOP!!!When you see such information, don’t accept it as something you can use for a research report. Ask yourself […]
Changing Density Units
Density is a type of conversion factor that compares the mass of a specific volume. The basic metric unit for mass is gram, g.The basic metric unit for length is meter, m.The basic metric unit for volume is L or m 3. Use the density of calcium, Ca, found at Dynamic Periodic TableConvert the units of […]
Hidden Colors
Paper Chromatography Chromatography means to write with colors. Chromatography is a chemical process of separating mixtures of colorants. The first video below demonstrates the following Paper Chromatography procedure steps. The video also has a craft idea for making a butterfly using the colored paper filter.The second video below provides information about paper chromatography as well as more […]
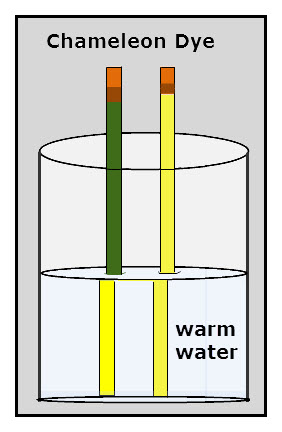
Chameleon Dyes
Chameleon Dyes Note: I am revising the following information. I had the opportunity to speak with one of the directors of LCR Hallcrest, a company that makes chameleon dyes. The pencils in the picture are called chameleon pencils. While chameleons change colors in response to different stimuli, such as temperature, emotions, and illness, the dye […]
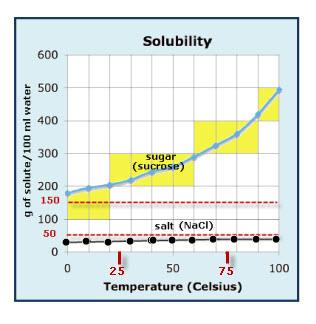
Solubility Graph
I am always searching for new, fun crystal growing recipes. Crystals of table sugar and table salt are favorites, mainly because the chemicals are so readily available. The solubility graph shown provides information about the amount of sugar and salt that can be dissolved at different temperatures. When you mix sugar and water, the mixture […]
Cross-Linked Polymers
Polymers are chemicals made up of repeating chemical units, called monomers. Polymerization is the process by which monomers attach to each other to form a chain made up of thousands of monomer units. *Styrofoam is the Dow Chemical’s brand name for foamed polystyrene. Cross-linking Single threads of fiber have more uses when twisted together to form rope […]
What is Pig Iron?
Pig iron is the intermediate product of removing native iron (metalic iron or iron alloy)) from its ore (iron compounds + impurities called slag). Iron Ore + heat——————> Pig Iron [native iron mixed with slag] Pig Iron + heat —————–> Native Iron + Slag (impurities from iron ore) What is Native Iron? To understand the previous statements, […]
How Many Atoms in A Mole?
A chemical mole is not an animal. A chemical mole is a unit of measuring the amount of chemical substances. For the problem shown, use this mole conversion factor: 1 mole = 6.o2 x 10 23 atoms The number 6.o2 x 10 23 is called Avagadro’s Number Calculate: 4.2 x 10 26 atoms of lithium (Li) are equal to how many […]
Thermochromic Ink
How Does Thermochromic Ink Work? Let’s start with the term thermochromic, which describes the ability of something to change color with changes in the temperature. Thus, when thermochromic ink is used on a material, the part where the ink is will change colors with fluctuations in temperature. So how do thermochromic inks change color? Thermochromic ink […]
Who Wants to Cook with Chemicals?
Let’s Use Our Hands to make a Chemical Mixture Homemade play dough is a great science experiment for young learners. But, more than learning about chemistry, this play dough activity connects to other curriculum, including math and art. But one of the best parts is that it is FUN! Even adults will want to get […]
- « Previous Page
- 1
- 2
- 3
- 4
- …
- 19
- Next Page »