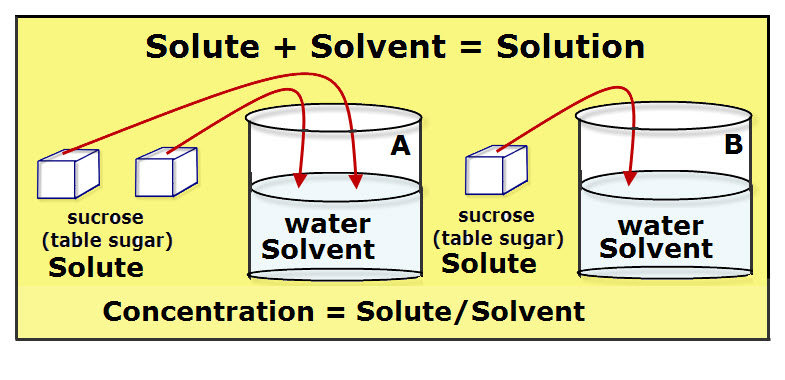
What is a Solution? How do You Saturated A Solution? A solution is a homogeneous mixture, meaning it is the same throughout. A solution can be made of more than two substance, with one of the substances being the solvent and the substances that dissolve in the solvent are called solutes. You make and use […]
Snowflakes
Six-Sided Snowflake Models The snow flake to the right gives the basic design of a six-sided snowflake. You can be as creative as you dare in making snowflake models. Just be sure they have six major sides. A paper snowflake can be cut from a round paper coffee filter. The procedure for folding and cutting […]
How Crystals Form
What You Need to Know: A crystal is a solid in which the particles are packed in an ordered, repeated pattern. Precipitation is the process by which a solid falls out of a solution. A precipitate is the solid that precipitates. An evaporite is a precipitate formed by evaporation. What Does This Mean? Crystals are […]