 Discovery Investigation: DNA Extraction
Discovery Investigation: DNA Extraction
1. Why is the banana smashed?
Smashing the banana breaks cell wall as well as cell membranes so that the nuclei are released.
2. What is the purpose of the dish washing liquid?
The membrane around cell membranes as well as the membrane around the nuclei contains material that dissolves in the soap. DNA is found in the nucleus of the cells. With the nuclear membrane broken, DNA is released. DNA is soluble in water, so it is part of the aqueous filtrate collected when the mushy banana mixture is filtered through cheese cloth.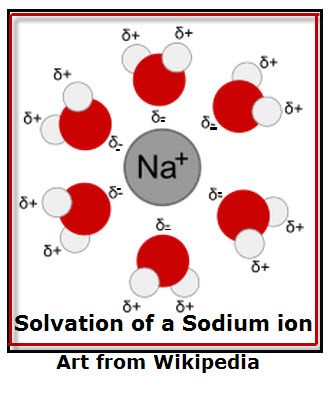
3. What is the purpose of the table salt?
Table salt dissolves in water forming sodium ions and chloride ions. The sodium ions are positively charged. Part of the DNA molecule is negatively charged so it is attracted to the sodium ions, but in the water, the two ions remain separated because of the solvation process.
4. What does solvation mean? How does solvation keep the DNA and sodium ions from combining?
Solvation, is the process by which the molecules of a solvent are attracted to solute ions. For example, sodium chloride dissociates (breaks into ions) when dissolved in water. As the ions diffuse (sprea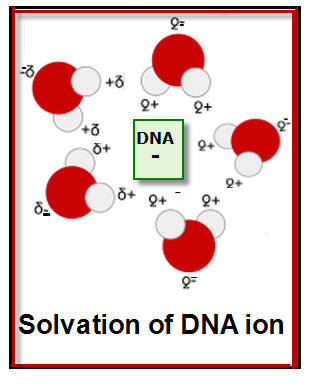 d out) in water, the separate ions become surrounded by water molecules.This is because water molecules are polar (have slightly positive charges on one side and a slightly negative charges on the opposite side). The charged water molecules keep ions (charged particles) from combining. Water forms a “fence” around the ions as shown in the diagram. Notice that the negative side of the water molecule (indicated by ? (delta sign) with two negative (-) charges points toward the positive sodium ion.
d out) in water, the separate ions become surrounded by water molecules.This is because water molecules are polar (have slightly positive charges on one side and a slightly negative charges on the opposite side). The charged water molecules keep ions (charged particles) from combining. Water forms a “fence” around the ions as shown in the diagram. Notice that the negative side of the water molecule (indicated by ? (delta sign) with two negative (-) charges points toward the positive sodium ion.
A comparable but reverse diagram and the positive part of the water molecules points toward the negative DNA ion.
5. Interface Method of Extracting DNA: What is the purpose of the alcohol?
Without the alcohol, the sodium and DNA ions could not join and precipitate (come out of solution). Alcohol is not as polar as water, so where the aqueous filtrate touches the alcohol layer, the sodium ion and DNA ion bonds (connect) and precipitate.
When the sodium and DNA molecules start precipitating more and more of these molecules start sticking together. It is almost as if the precipitated molecules grab onto other sodium and DNA –pulling them out of solution. This is called flocculation.
As the slimy looking combined Na DNA molecules clump together they push up into the alcohol. This is because they are not soluble in the alcohol. Unlike being in water, where the Na and DNA ions are solvated by water molecules, the two ions bond remain bonded to each other in the alcohol. The alcohol molecules float around the precipitated clump of Na DNA.
5. Mixture Method of Extracting DNA: What would happen if the alcohol and aqueous filtrate were mixed together?
If there is about 1 part isopropyl alcohol (rubbing alcohol) and 2 parts aqueous filtrate, The sodium + DNA molecule will not dissolve in it. There will be more DNA precipitate in this mixture but there will also be other precipitates. This means the DNA extract will not be very pure.
With the interface method (interface is the boundary between the alcohol layer and the aqueous filtrate layer) the DNA precipitates at the boundary and tends to be somewhat more pure. The problem is that only a small amount of the DNA is recovered.


